POVAL Characteristics
Water solubility
POVAL is a water-soluble polymer and can be dissolved in water at the desired ratio.POVAL aqueous solutions have a suitable viscosity and can be coated on a variety of objects to form a surface membrane when dried.
Solvent Resistance
POVAL will only dissolve in certain solvents, such as DMSO (dimethylsulfoxide), and water. It will not dissolve and remains stable in the presence of organic solvents, animal and plant oils, and grease.
Adhesive Strength
POVAL has hydroxyl groups, so it exhibits a powerful adhesive force to hydrophilic surfaces, such as paper or wood.
Chemical Reactivity
POVAL has hydroxyl groups, so it can be reformed through acetalization reactions and reactions with boric acid and borax among others.
Membrane Formation
POVAL has excellent membrane formation, high film transparency, high strength, excellent gas barrier, clouding prevention, and anti-static electricity.
Surface Activation
POVAL has hydrophilic hydroxyl groups and hydrophobic acetate groups, so it exhibits excellent surface activation, emulsion, and dispersion properties.
Stability
POVAL is a stable polymer that will not change or deteriorate in general environments
Effects of the Degree of Polymerization and Degree of Hydrolysis
POVAL’s characteristics are affected by the degree of polymerization and the degree of hydrolysis.
The following table shows the affect of the degree of polymerization and degree of hydrolysis on POVAL’s basic characteristics.
| Characteristic | Degree of Polymerization | Degree of Hydrolysis | ||
| Low | High | Low | High | |
| Low Degree of Polymerization Products |
High Degree of Polymerization Products |
Partially Hydrolyzed Type | Fully Hydrolyzed Type | |
| Viscosity of solution | ||||
| Viscosity stability at low temperature |
||||
| Membrane strength | ||||
| Water solubility | ||||
| Water resistance | ||||
| Emulsifying ability | ||||
The greater the number of arrows shown, the greater the affect.
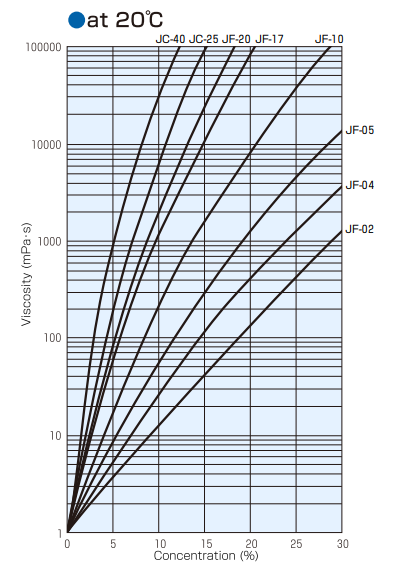
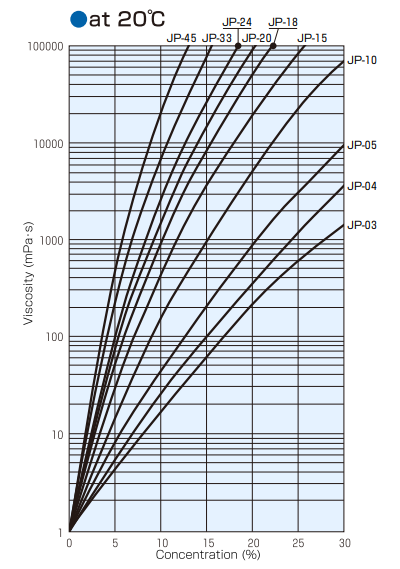
Viscosity of Solution
Relationship Between Aqueous Solution Concentration,Temperature, and Viscosity (Type B Viscometer)
Aqueous Solution Stability
Aqueous Solution Surface Tension
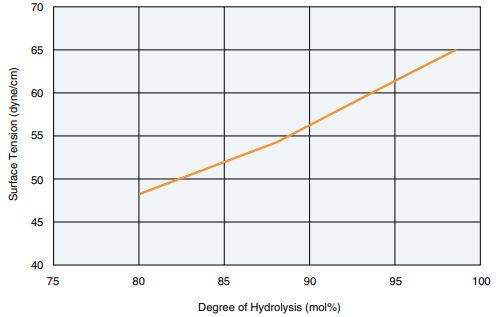
- Degree of polymerization: 1700
- Aqueous solution concentration: 0.2%
- Measurement: Wilhelmy method
The POVAL aqueous solution surface tension is greater the higher the degree of hydrolysis,and lower the lower the degree of hydrolysis.
Membrane Properties
Membrane Strength
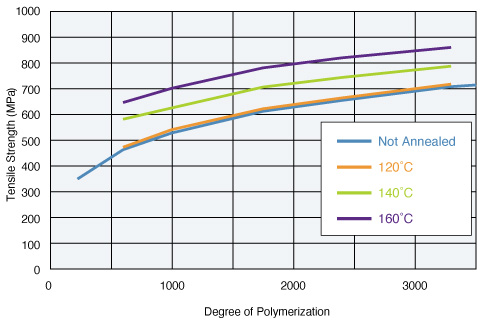
- Degree of hydrolysis: 98.5 mol%
- Tension speed: 50 mm/min (Measured at 20˚C and 65%RH.)
The higher the degree of polymerization, the stronger the POVAL membrane. When thermal processing is performed, crystallization progresses and further increases the membrane strength.
Membrane Water Resistance
Relationship Between the Degree of Hydrolysis and the Percentage of Insoluble Matter
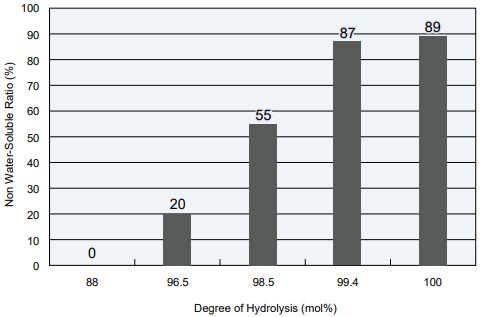
- POVAL: degree of polymerization: 1700
- Film: Membrane with a 100 µm thickness formed by drying a flow of POVAL aqueous solution at room temperature.
- Measurement conditions: Let soak for 24 hours in water at 40ºC.
The higher the degree of hydrolysis, the stronger the water resistance of the POVAL membrane.
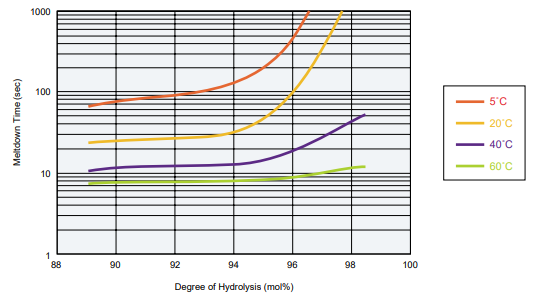
Film Solubility
POVAL Degree of Polymerization: 600
POVAL Degree of Polymerization: 1700
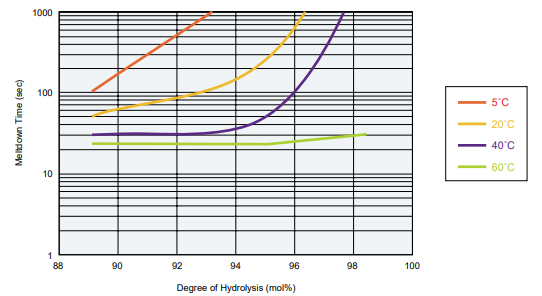
- Film: Membrane with a 40 µm thickness formed by drying a flow of POVAL aqueous solution at room temperature.
- Measurement conditions: Soak the film in waters of various temperatures and measure the meltdown time.